 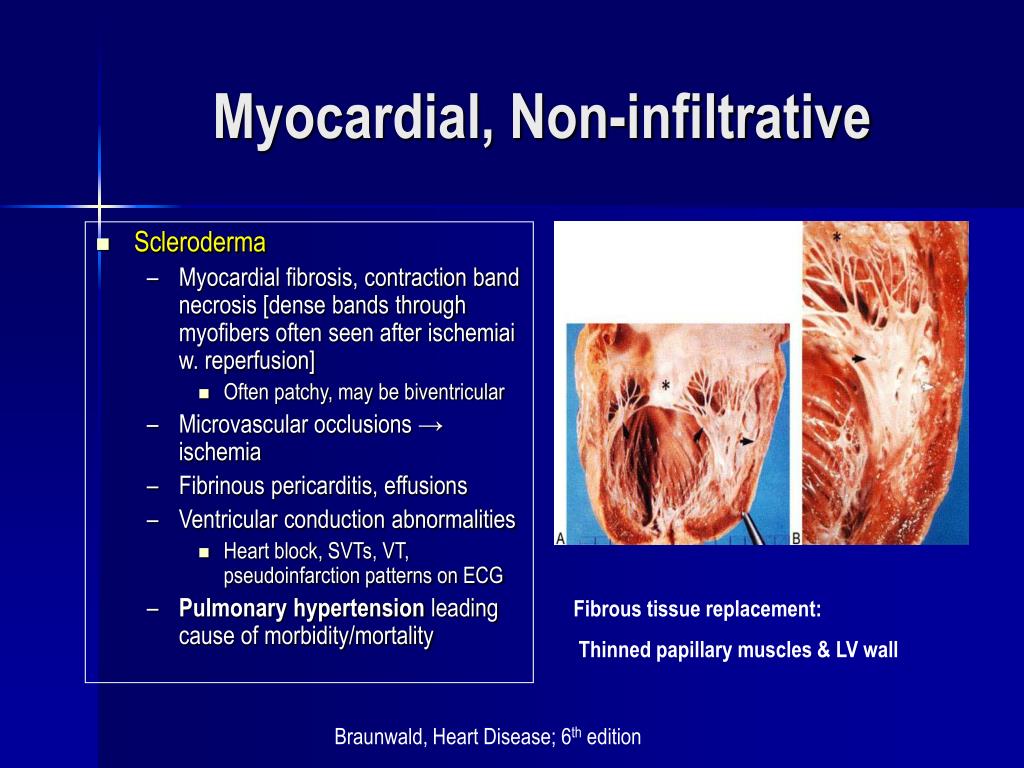
The obliteration of vessel lumen by neutrophil-platelet aggregates is associated with release of vasoconstrictors and inflammatory mediators (in brown). Reperfusion injury: the principal determinants of this phenomenon are represented by neutrophils (in green), endothelin-1, thomboxane-A2, and platelets (in yellow). Interstitial myocardial oedema compresses capillaries and small arterioles, further decreasing flow through these dysfunctional vessels, whereas sodium and calcium overload explains myocardial cell swelling. Ischaemic injury: it depends on duration and extent of ischaemia and is characterized by severe capillary damage, endothelial protrusions, and blebs that block the capillary lumen, and endothelial gaps with extra vascular erythrocytes (in red). There are four interacting mechanisms involved in the pathogenesis of coronary microvascular obstruction in humans: ischaemia-related injury, reperfusion-related injury, distal embolization, and individual susceptibility (both genetic and due to pre-existing coornary microvascular dysfunction) of the microcirculation to injury. 5, 7 Furthermore, previous studies indicate that abnormal non-endothelium-dependent microvascular dilatation appears to be involved in functional and structural alterations that lead to impaired coronary flow reserve (CFR) with aging, hypertension, diabetes, dyslipidaemia, insulin resistance, and chronic inflammatory diseases. Traditional and non-traditional risk factors play a role in epicardial and microvascular endothelial-dependent dysfunction, specifically in the high-risk subset with STEMI ( Figure 1). Mechanisms of coronary microvascular dysfunction and obstruction Pre-existing coronary microvascular dysfunction In this article, we review mechanisms, diagnosis, and prognosis of CMVO in acute STEMI, also addressing the prevention and treatment of CMVO by highlighting the need for an integrated approach in different time windows. 5, 6 Hence, at the time of reperfusion, patients with pre-existent microvascular dysfunction will benefit less from prompt reopening of the epicardial vessel, thus underscoring that, preserving a normal microvascular function before acute coronary occlusion, is a crucial target of preventive therapies for CMVO similarly to those aiming at restoring flow in the microcirculation during primary PCI and thereafter in the coronary care unit (CCU). Coronary microvascular dysfunction has been shown to increase the risk of cardiovascular events regardless of the epicardial disease status. To this regard, an unmet need is to address the coronary microvascular functional and structural obstruction (CMVO), which occurs frequently even after prompt epicardial recanalization of the infarct-related artery. 3, 4 Additional strategies are needed to reduce in-hospital mortality in this population and attention has to be turned to the development of systems addressing the continuum of STEMI care, from symptom onset through return to community. 2 However, although national door-to-balloon times have improved significantly over the last years for patients undergoing primary PCI, in-hospital mortality has remained virtually unchanged. Accordingly, reducing time to treatment and maximizing myocardial salvage-in keeping with the mantra that ‘time is muscle’-represents a major challenge in the management of STEMI. 1 The goal of reperfusion therapy with fibrinolytic drugs or primary percutaneous coronary intervention (PCI) is to restore blood flow to ischaemic, but still viable, myocardium, and reduce infarct size (IS). ST-segment elevation myocardial infarction (STEMI) usually results from acute thrombotic occlusion of a coronary artery and is a leading cause of death. Microvascular dysfunction, Microvascular obstruction, Acute myocardial infarction Introduction 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
